C1 Structure and properties
Simple molecular substances consist of molecules in which the atoms are joined by strong covalent bonds. However, the molecules are held together by weak forces so these substances have low melting and boiling points. They do not conduct electricity.
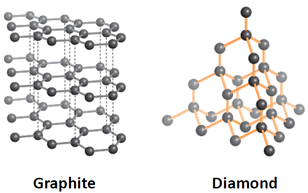
Giant covalent structures contain many atoms joined together by covalent bonds to form a giant lattice. They have high melting and boiling points. Graphite and diamond have different properties because they have different structures. Graphite conducts heat and electricity well because it also has free electrons.
Giant covalent structures contain many atoms joined together by covalent bonds to form a giant lattice. They have high melting and boiling points. Graphite and diamond have different properties because they have different structures. Graphite conducts heat and electricity well because it also has free electrons.
Key Facts in this topic |
Test yourself! |
