C2.1 Structure and bonding
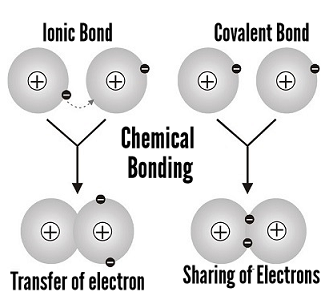
Ions are electrically charged particles formed when atoms lose or gain electrons. They have the same electronic structures as noble gases.
A covalent bond is a strong bond between two non-metal atoms. It consists of a shared pair of electrons. A covalent bond can be represented by a straight line or dot-and-cross diagram.
A covalent bond is a strong bond between two non-metal atoms. It consists of a shared pair of electrons. A covalent bond can be represented by a straight line or dot-and-cross diagram.
Key Facts in this topic |
Test yourself! |
