C1 Reaction of acids and Electrolysis
Acids have a pH of less than 7. Bases have a pH of more than 7. When bases are dissolved in water, they are known as alkalis. Salts are made when an acid reacts with a base, carbonate or metal. The name of the salt formed depends on the metal in the base and the acid used. For example, salts made using hydrochloric acid are called chlorides.
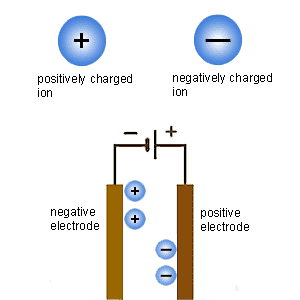
Electrolysis is the process by which ionic substances are broken down into simpler substances using electricity. During electrolysis, metals and gases may form at the electrodes.
Electrolysis is the process by which ionic substances are broken down into simpler substances using electricity. During electrolysis, metals and gases may form at the electrodes.
Key Facts in this topic |
Test yourself! |
